Introduction
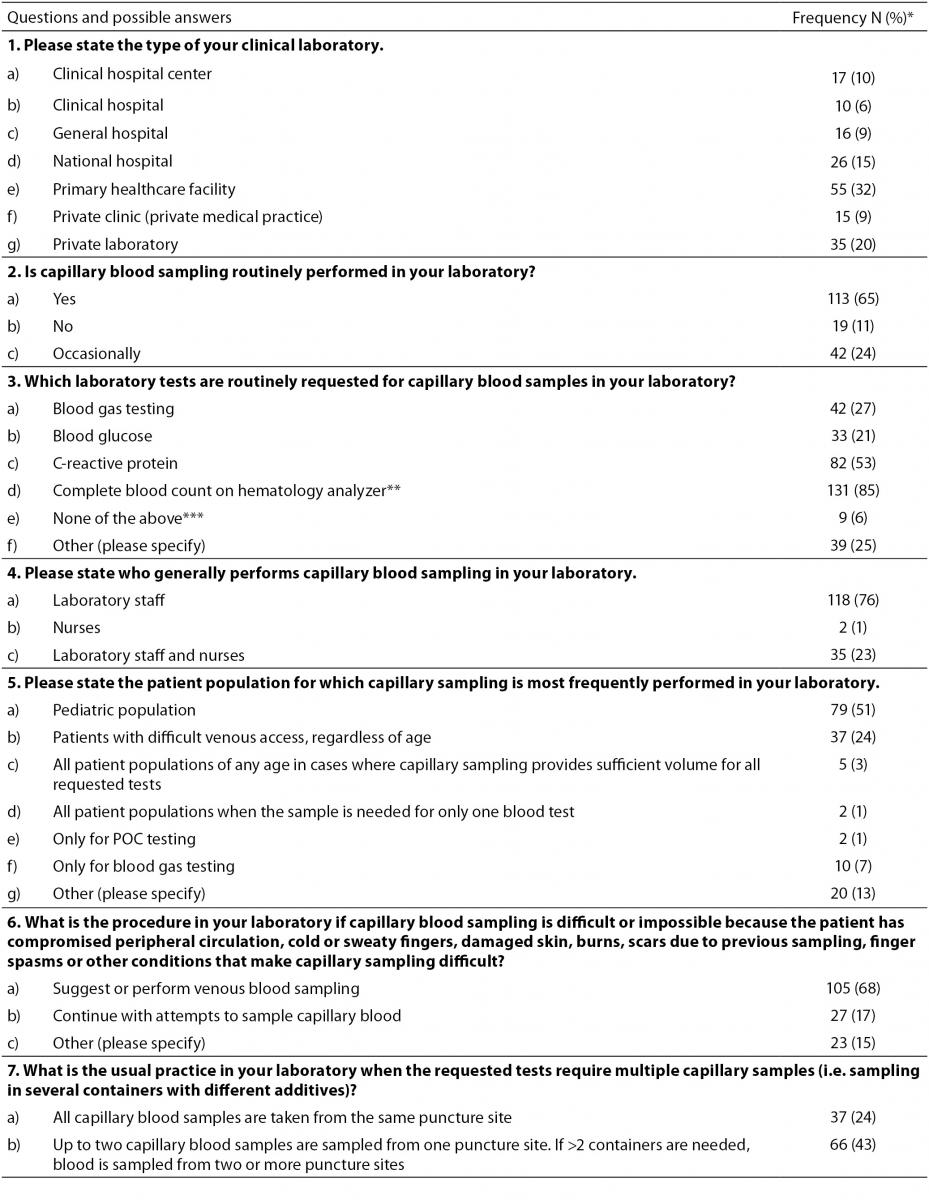
Capillary blood sampling allows much smaller blood volumes to be drawn in a much less invasive manner than venous sampling. Capillary blood samples are primarily arterial blood, though they also contain unknown proportions of blood from venules, arterioles, and capillaries, as well as from interstitial and intracellular fluid (1). Together with technological advances that allow multiple blood tests to be performed quickly and easily with small sample volumes, capillary sampling is helping to drive the widespread use of point-of-care (POC) diagnostics. POC analyzers are meant to allow clinical staff, rather than laboratory staff, to perform a variety of tests quickly and easily in the ward rather than in the laboratory (2).
Although capillary blood sampling has several advantages over venous sampling, it also carries greater risk of giving incorrect test results. This is because hemolysis and lipemia cannot be detected in capillary-sampled blood, and both can significantly affect test results. In addition, analyte concentrations can differ between capillary and venous blood, potentially invalidating standard reference ranges based on venous blood. Indeed, differences between the two sampling methods mean that clinicians must be careful to perform longitudinal testing using only one sampling type. These caveats for capillary testing highlight the need for adequate training of testing staff, yet capillary sampling, testing, and analysis of results is usually performed by non-laboratory personnel.
In addition to these procedural disadvantages, capillary blood sampling is unsuitable for patients who are dehydrated or edematous or who have poor blood circulation (3); these groups may be quite prevalent among inpatient populations and in emergency departments. In addition, capillary sampling is increasingly used to perform numerous blood tests, particularly for paediatric patients. These tests include complete blood count (CBC), blood gases and acid-base status (ABS) with or without electrolytes, blood glucose, C-reactive protein (CRP) and bilirubin. Performing multiple tests often requires drawing multiple samples that together may make up a relatively large volume, which may create problems for analysis. One is that larger blood samples can contain appreciable proportions of interstitial or intercellular fluid, changing the blood composition and rendering standard reference ranges unsuitable. Another problem is that risk of hemolysis is significantly greater for larger samples.
Thus, obtaining reliable laboratory test results requires adequate capillary blood sampling. Extensive, evidence-based guidelines for POC testing and diagnostics have been published in the Laboratory Medicine Practice Guidelines (4), requirements on quality is part of ISO standards (5) and document POCT07-P of the Clinical and Laboratory Standards Institute (CLSI) (6), and in relevant research articles (4,7). Procedure guidelines for capillary blood samples have been published in two documents: CLSI document GP42-A6 (3) and World Health Organization (WHO) Guidelines on Drawing Blood (8). CLSI and WHO guidelines recommend capillary sampling in newborns and children and they indicate that such sampling can in principle be used for adults, with the CLSI guidelines further defining when such sampling should be performed in adults. Adults for whom capillary sampling is recommended are elderly patients and those with severe burns, extreme obesity, or susceptibility to thrombosis; patients scheduled to receive intravenous therapy, regardless of their age; patients with vessel walls that may easily rupture; and individuals who perform glucose self-testing at home.
Guidelines from the CLSI (3) and WHO (8) clearly define all the steps in capillary blood sampling, including recommendations for choosing sampling sites and deciding the order of draw. The guidelines also prescribe actions to take if blood stops flowing, and whether to persist with attempts to sample capillary blood or switch to venous collection in the event of complications.
The good laboratory practice standards issued by the Croatian Chamber of Medical Biochemists in 2005 (9) provide no answers to these questions, raising the possibility that capillary sampling in Croatian laboratories does not comply with CLSI and WHO guidelines. Therefore, we surveyed more than 200 clinical laboratories throughout the country in order to assess the prevalence of capillary sampling for different patient populations and to assess compliance of protocols and procedures with international guidelines. The results may help identify which steps of capillary blood sampling require improvement in Croatia.
Materials and methods
Questionnaire
An online questionnaire entitled “Capillary sampling – advances and limitations” was created using web service SurveyMonkey (Palo Alto, CA, United States) and sent in October 2013 by email to all laboratories that routinely participate in the Croatian National External Quality Assessment program. This program, run by the Croatian Centre for Quality Assessment in Laboratory Medicine (CROQALM), is obligatory for all licensed medical laboratories in Croatia.
The questionnaire comprised 10 questions intended to provide data on the type of clinical laboratory, the types and proportions of laboratory tests performed from capillary samples, and the primary patient population(s) for which capillary sampling was used. The questionnaire also asked about practices and protocols for capillary sampling, as well as the existence and source of reference intervals used to interpret test results.
Statistical analysis
Responses to the survey were shown in terms of absolute numbers and percentages of respondents. Frequencies of responses were compared using the test for comparison of proportions. Statistical analysis was performed using the free software MedCalc 13.1 (Ostend, Belgium). P < 0.05 was defined as the threshold of significance.
Results
Of the 204 medical laboratories invited to participate, corresponding to all the licensed laboratories in Croatia, 174 (85%) completed the questionnaire. Most respondents were located within primary healthcare facilities or were private laboratories (Table 1). Of the 174 respondents, 113 reported performing capillary sampling routinely, 40 reported performing it occasionally, and 21 only in special cases. Two laboratories indicated on question 3f that they perform capillary sampling for CBC and CRP tests on rare occasions. We contacted these laboratories for more information, based on which we decided to include them among the laboratories that occasionally perform capillary sampling. In the end, the total number of laboratories performing capillary sampling either routinely or occasionally was 155 (89% of respondents).
Table 1. Survey of capillary blood sampling practices in clinical laboratories in Croatia.
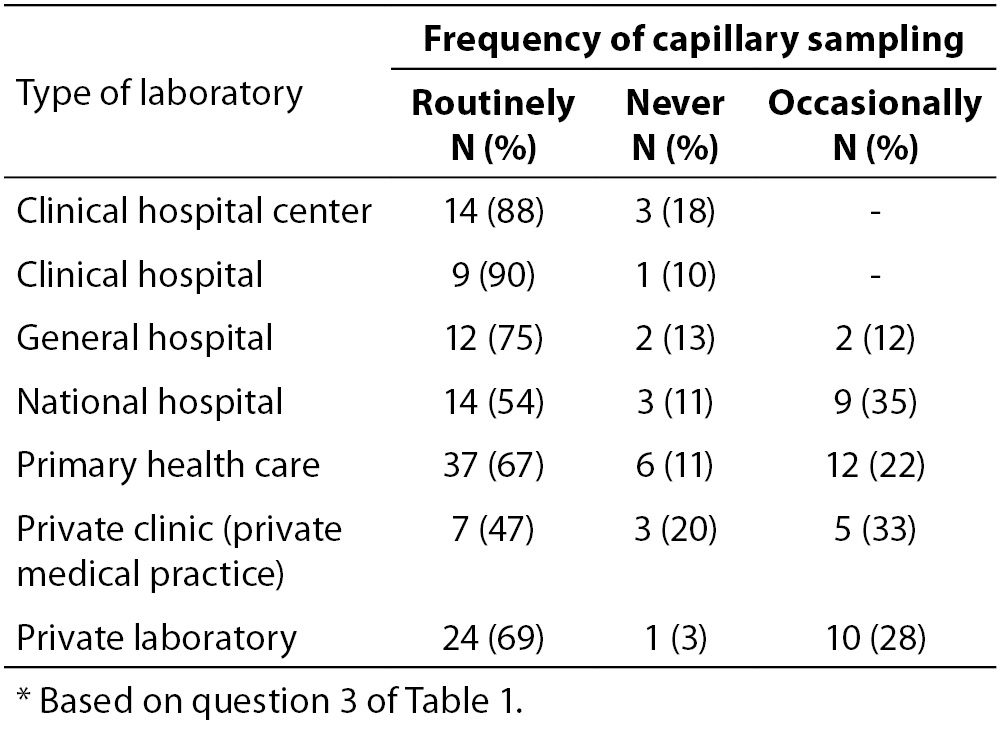
The proportions of laboratories performing capillary sampling routinely, occasionally or never did not vary significantly among the various types of laboratories in our sample (Table 2). Most laboratories use capillary sampling to determine CBC on a hematology analyzer, while smaller proportions of laboratories use it to test for CRP, gases and glucose (Table 1, question 3). A similar proportion of respondents uses capillary sampling to determine blood gases (27%) and blood glucose (21%; P = 0.268). Only 6% of respondents do not perform capillary sampling for these four most frequent laboratory tests. One-quarter of respondents perform capillary sampling for additional laboratory tests, independently of or in combination with the four mentioned on the survey. Additional tests filled in by respondents (question 3f) include prothrombin time, bilirubin, liver function enzymes, urea, creatinine, and iron.
Our respondent sample included similar proportions of laboratories that use capillary blood sampling for 1, 2 or ≥3 laboratory tests (Figure 1). After grouping the latter two categories, we found that a significantly higher proportion of laboratories perform capillary sampling for ≥ 2 tests (69%) than for only 1 test (31%, P < 0.001).
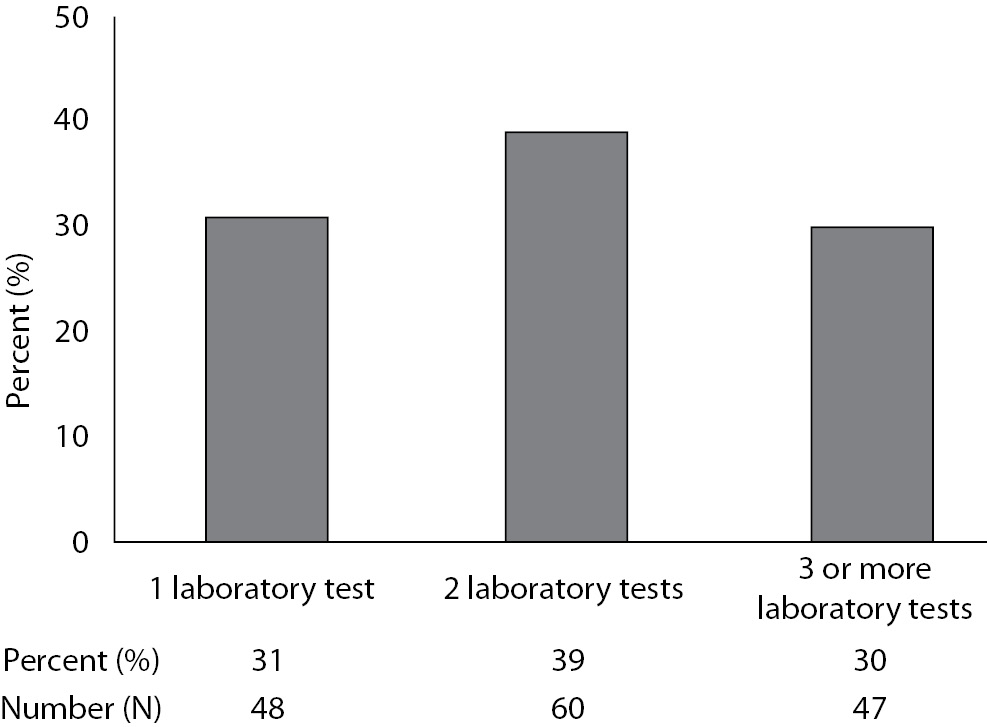
Figure 1. Numbers of tests routinely performed on capillary blood samples across 155 clinical laboratories in Croatia. Data come from responses to question 3 in Table 1.
Most respondents (76%) indicated that laboratory staff, not physicians or nurses, are responsible for sampling capillary blood. Only 1% of respondents reported that nurses are responsible for capillary sampling, while the remaining 23% reported that either laboratory staff or nurses perform it.
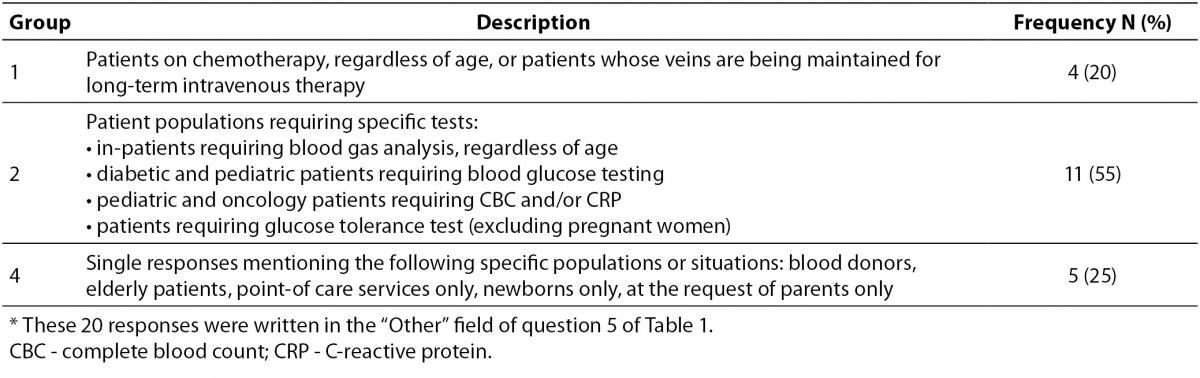
One-quarter of respondents indicated that they use capillary sampling in all individuals in whom venous access is difficult, regardless of age (Table 1, question 5). Half of the respondents indicated that they use it primarily for children. Twenty laboratories reported using capillary sampling primarily for “other” populations not listed (question 5g), which we were able to divide into three groups (Table 3). Just over half (55%) of the 20 responses referred to group 2 (patients requiring specific tests), which was not significantly higher than the percent of responses belonging to group 1 (20%, P = 0.050) or group 3 (25%, P = 0.107).
Most laboratories (68%) reported that when capillary sampling proves difficult or impossible, they suggest or perform venous sampling (question 6, Table 1). This proportion is significantly larger (P < 0.001) than the proportion that insist on capillary blood sampling (17%) or that follow another course of action (15%); the proportions for these latter two groups are similar (P = 0.745). Other courses of action (with % proportions) were as follows: the laboratory has yet to encounter this problem and so does not have a defined practice (29%); the person sampling would warm the hand and try again (15%), consult with a physician and try again (10%), attempt again at another puncture site (10%), or request venous or arterial sampling (9%); the laboratory takes a specific action depending on the requested test and the patient’s condition.
Of the 155 laboratories that occasionally or regularly use capillary sampling, 48 (31%) perform only single sampling for one laboratory test (Figure 1; Table 1, question 3). The remaining 107 laboratories perform sampling for two laboratory tests (N = 60) or for ≥3 tests (N = 47). Both the entire set of 155 laboratories and the subset of 107 gave similar responses to the question of how multiple samples were prepared in different containers (Figure 2; Table 1, question 7).
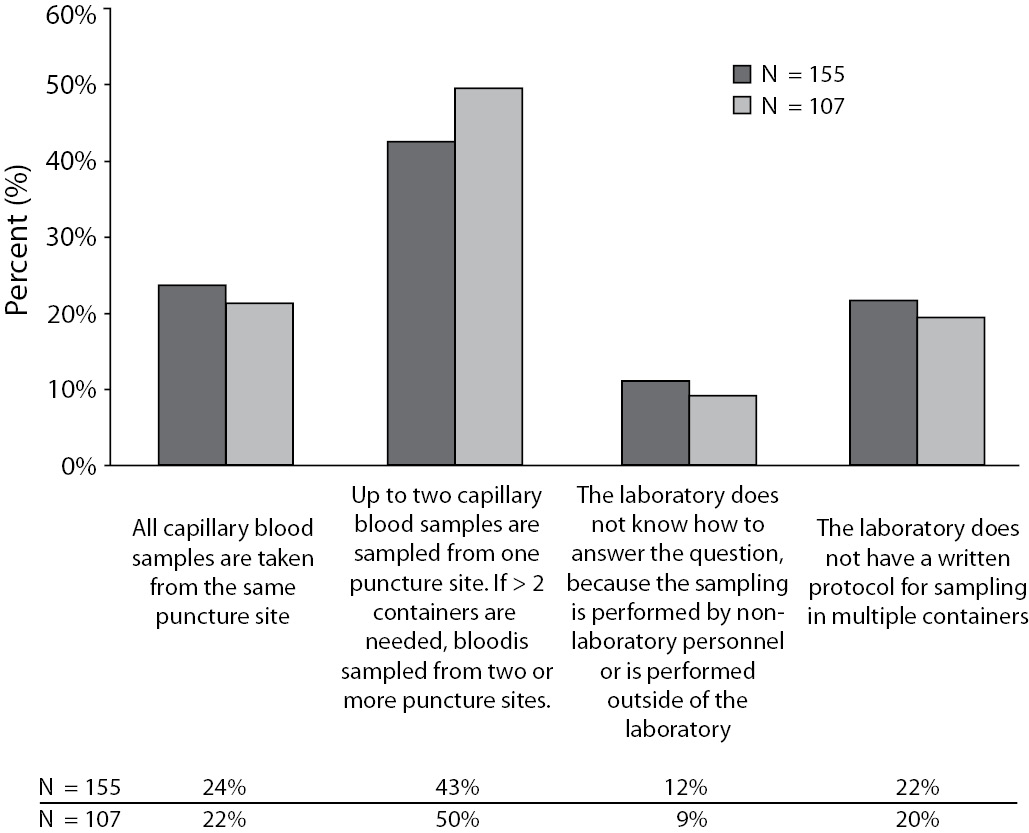
Figure 2. Comparison of how multiple sampling of capillary blood is performed in both the total set of 155 laboratories performing capillary blood samping and the subset of 107 laboratories routinely performing multiple capillary sampling. Data come from question 7 in Table 1.
Table 2. Frequency of capillary blood sampling by type of clinical laboratory in Croatia.*
Of the laboratories that perform capillary blood sampling (Table 1, question 8), 78% have some kind of written protocol for the procedure, but only 30% have a written protocol that includes guidelines on order of draw for multiple sample collection.
Nearly all laboratories (92%) use the reference intervals for laboratory tests based on capillary samples that are endorsed by the Croatian Chamber of Medical Biochemists in its document “Harmonization of Laboratory Tests” (Table 1, question 9) (9). Reference intervals for tests not regulated by this harmonization document are taken from declarations of test manufacturers or other professional sources. Only 3% of laboratories reported verifying the applicability of reference intervals taken from other sources. An equally small proportion (3%) uses the same reference intervals for venous, arterial or capillary blood samples.
Table 3. Patient populations in Croatia for which capillary blood sampling is primarily used.*
Most laboratories (88%) report never performing arterialisation prior to capillary blood sampling (Table 1, question 10). Most of the remaining laboratories perform it primarily for blood gas testing, when fingers are cold or when blood circulation is poor. Only 1% of laboratories responded that they use only arterialized capillary blood samples.
Discussion
This is the first study to assess the frequency and practices of capillary blood sampling in Croatia, based on a sample of 85% of all medical laboratories in the country. Among the 174 respondents, 90% perform capillary sampling regularly or occasionally as part of their routine practice. Our sample contained primarily publicly funded laboratories, but 29% were private, a proportion higher than the 7% sampled in a previous nationwide survey study of laboratory practice (10). Our results suggest that capillary sampling is prevalent across all patient populations, regardless of age, and across medical laboratories at all types of healthcare institutions.
To ensure that capillary samples are of sufficient quality, CLSI and WHO guidelines recommend that laboratories develop a written protocol for capillary sampling, and that the protocol specify the order of draw. This is important because the proportions of arterial, venous and capillary blood change over time, as does the ratio of interstitial to intercellular fluid. This can significantly affect the results of laboratory analyses. This contrasts with venous sampling, where order of draw serves mainly to minimize carryover of additives from one sample to another (1,3,8,11). Of the 107 laboratories in our sample that perform capillary sampling for ≥ 2 laboratory tests, with different tests requiring containers with different additives, half have a written protocol for capillary sampling that fails to specify order of draw. This likely reflects the fact that order of draw is absent from the good laboratory practice guidelines of the Croatian Chamber of Medical Biochemists (9).
Although capillary sampling for POC diagnostics is designed to be performed by non-laboratory personnel with minimal training, laboratory technicians at many medical facilities continue to bear the responsibility for laboratory diagnostic tests performed outside the laboratory (12). Therefore, POC instruments at many facilities are located within the medical laboratories. This likely explains why nurses perform capillary sampling in only 1% of our laboratories, while a combination of laboratory staff and nurses perform it in 23% of laboratories. Although nurses are expected to be able to perform both capillary and venous sampling (13-15), laboratory staff is responsible for defining the limitations of the procedure and for standardizing the sampling protocol throughout the medical facility. Our results indicate that many laboratories lack such written protocols, or their written protocols do not specify blood order of draw, highlighting the need for greater efforts to standardize procedures. Ultimately, national guidelines are needed to ensure compliance by individual laboratories.
More evidence for lack of standardization was observed in responses to the question of what actions laboratories take when capillary sampling becomes challenging or fails entirely. Although the majority reported that they would ask for a venous sample, a significant proportion would persist in their effort to collect a capillary sample. The WHO recommends that capillary sampling be abandoned after two unsuccessful attempts (8). Laboratories also differ in their procedures for collecting multiple capillary samples from the same patient into multiple containers. Only approximately half the laboratories reported that they collect no more than two capillary samples from the same prick site; if more samples are needed, another puncture is made. The remaining laboratories collect all samples for requested tests from one puncture site, or they have no clear rules about multiple sampling, or they leave decisions up to the best judgment of whoever is performing the sampling, especially at facilities where the sampling is performed outside the laboratory. Clear guidelines for multiple sampling are particularly important for cancer patients on chemotherapy and for patients in the emergency department. Many of these patients are dehydrated, have poor peripheral blood circulation or edemas, or have multiple wounds from previous capillary sampling at the recommended puncture sites (fingers and heel). In all these situations, CLSI and WHO guidelines recommend against capillary sampling (3,8). However, strict adherence to these guidelines is often impossible if the patient is a child or newborn in critical condition, since venous sampling is not an option. This highlights a discrepancy between guidelines and clinical reality that should be addressed at the national level in Croatia and ultimately at the international level.
CLSI guidelines recommend that only arterialized capillary blood samples be used, while WHO guidelines do not go so far and recent studies suggest this may be unnecessary for certain tests (3,8,16,17). Nearly 90% of the laboratories in our sample reported that they never perform arterializations prior to capillary sampling. A small proportion (8%) of laboratories use arterialized capillary blood samples only when testing blood gases. These results are similar with the results of a recently published survey study about procedures for acid-base testing (18).
Our findings on the prevalence of capillary sampling in Croatia and the range of tests for which it is used suggest that it is driving an increase in POC diagnostics in the country, consistent with what is happening in other countries. At the same time, many Croatian laboratories use sampling practices that deviate substantially from CLSI and WHO guidelines. This highlights the importance of defining and standardizing practices at the national level and aligning these practices with international guidelines, particularly with respect to developing written sampling protocols, specifying order of draw for multiple samples, and adopting appropriate actions if sampling becomes difficult or fails.
Conclusion
This national survey of capillary sampling in Croatia shows that the procedure is common throughout the country, independent of the type of medical laboratory or patient group. The routine use of capillary sampling in Croatia tracks with the global increase in POC diagnostics. Capillary sampling in Croatia is often used in all patient populations, although it is most often used in children. Most laboratories use reference intervals consistent with national guidelines. However, capillary sampling practices in Croatian laboratories deviate in several respects from international guidelines. The most serious deviations are that many laboratories (1) lack written protocols for capillary sampling or the protocols exist but do not specify order of draw for multiple sampling, (2) lack clear guidelines for actions to take when capillary sampling is difficult or unsuccessful, (3) lack guidelines about when to perform capillary sampling from one puncture site or multiple sites, (4) do not perform arterialisation before capillary sampling or (5) do not perform capillary blood sampling themselves, instead leaving it to nurses. Developing written rules and procedures to cover these issues, preferably at the national level, may significantly help standardize capillary sampling procedures around the country, while also aligning them with international guidelines.
Acknowledgments
The author is grateful to the participating clinical laboratories and their employees who filled out the questionnaire, and to the Croatian Society of Medical Biochemistry and Laboratory Medicine for providing access to the CLSI guidelines on capillary sampling. The author also thanks Professor Lidija Bilić-Zulle, PhD for assistance in preparing the questionnaire and Professor Ana-Maria Šimundić, PhD for critical comments on the manuscript.


