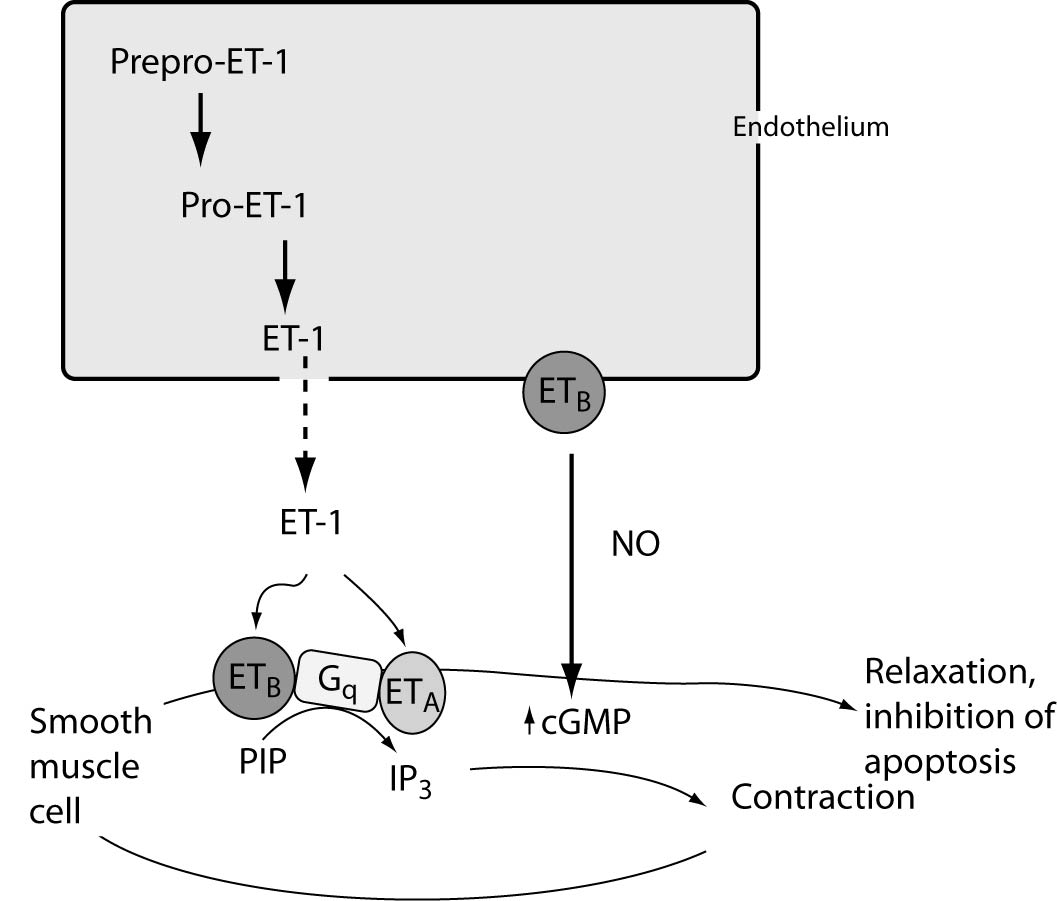
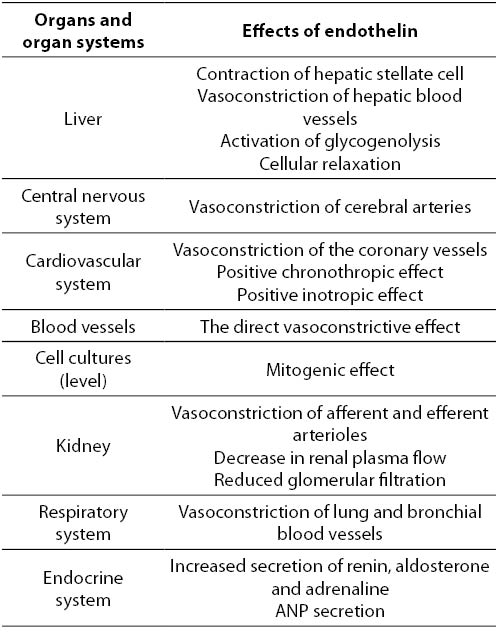
References
1. Yanagisawa M, Kurihara H, Kimura S, Tomobe Y, Kobayashi M, Mitsui Y, et al. A novel potent vasoconstrictor peptide produced by vascular endothelial cells. Nature 1988;332:411-5.
2. Kawanabe Y, Nauli SM. Endothelin. Cell Mol Life Sci 2011;68:195-203.
3. Simonson MS. Endothelins: Multifunctional renal peptides. Physiol Rev 1993;73:375-411.
4. Piechota A, Polanczyk A, Goraca A. Role of endothelin-1 receptor blockers on hemodynamic parameters and oxidative stress. Pharmacol Rep 2010;62:28-34.
5. Firth JD, Ratclife PJ. Organ distribution of the three rat endothelin messenger RNAs and the effects of ischemia on renal gene expression. J Clin Invest 1992;90:1023-31.
6. Thorin E, Clozel M. The cardiovascular physiology and pharmacology of endothelin-1. Adv Pharmacol 2010;60:1-26.
7. Opgenorth TJ, Wu-Wong JR, Shiosaki K. Endothelin-converting enzymes in rat lung. Biochem Biophys Res Commun 1990;171:1291-6.
8. Shimada K, Takahashi M, Tansawa K. Cloning and functional expression of endothelin-converting enzyme from rat endothelial cells. J Bol Chem 1994;269:18275-8.
9. Xu D, Emoto N, Giaid A, Slaughter C, Kaw S, deWit D, et al. ECE-1: a membrane-bound metalloprotease that catalyzes the proteolytic activation of big endothelin-1. Cell 1994;78:473-85.
10. Emoto N, Yanagisawa M. Endothelin-converting enzyme-2 is a membrane-bound, phosphoramidon-sensitive metalloprotease with acidic pH optimum. J Biol Chem 1995;270:15262-8.
11. Hasegawa H, Hiki K, Sawamura T, Aoyama T, Okamoto Y, Miwa S, et al. Purification of a novel endothelin-converting enzyme specific for big endothelin-3. FEBS Lett 1998;428:304-8.
12. Meidan R, Klipper E, Gilboa T, Muller L, Levy N. Endothelin-converting Enzyme-1, Abundance of Isoforms a–d and Identification of a Novel Alternatively Spliced Variant Lacking a Transmembrane Domain. J Biol Chem 2005;280:40867–74.
13. Marsen T, Schramek H, Dunn MJ. Renal action of endothelin: Linking cellular signaling pathways to renal disease. Kidney Int 1994;45:336-44.
14. Cerdeira AS, Brás-Silva C, Leite-Moreira AF. Endothelin-converting Enzyme Inhibitors: their Application in Cardiovascular Diseases. Rev Port Cardiol 2008;27:385-408.
15. Ikeda S, Emoto N, Alimsardjono H, Yokoyama M, Matsuo M. Molecular isolation and characterization of novel four subisoforms of ECE-2. Biochem Biophys Res Commun 2002;293:421-6.
16. Lorenzo MN, Khan RY, Wang Y, Tai SC, Chan GC, Cheung AH, et al. Human endothelin converting enzyme-2 (ECE2): characterization of mRNA species and chromosomal localization. Biochim Biophys Acta 2001;1522:46-52.
17. Kisanuki YY, Emoto N, Ohuchi T, Widyantoro B, Yagi K, Nakayama K, et al. Low blood preasure in endothelial cell-specific endothelin 1 knockout mice. Hypertension 2010;56:121-8.
18. Schini VB, Hendrickson H, Heublein DM, Burnett JC. Jr. Thrombin enhances the release of endothelin from cultured porcine aortic endothelial cells. Eur J Pharmacol 1989;165:333-4.
19. Kurihara H, Yoshizumi M, Sugiyama T, Takaku F, Yanagisawa M, Masaki T, et al. Transforming growth factor-beta stimulates the expression of endothelin mRNA by vascular endothelial cells. Biochem Biophys Res Commun 1989;159:1435-40.
20. Rubanyi GM, Vanhoutte PM. Hypoxia releases a vasoconstrictor substance from the canine vascular endothelium. J Physiol 1985;364:45-56.
21. Fujikawa H, Kawasaki K, Sekiguchi H, Mito H, Hayashi Y, Funayama H, et al. Endothelin-1 production in coronary circulation in a new canine model of coronary thrombosis. Cardiovasc Res 1996;32:604-12.
22. Nakaki T, Ohta M, Kato R. Inhibition by prostacyclin and carbacyclins of endothelin-induced DNA synthesis in cultured vascular smooth muscle cells. PLEFA 1991;44:237-9.
23. Alberts B, Bray D, Lewis J, Raff M, Roberts K, Watson JD. Cell signalling. In: Molecular biology of the cell. New York and London: Garland Publishing Inc; 1994. p. 721-87.
24. Karne S, Jayawickreme CK, Lerner MR. Cloning and characterization of an endothelin-3 specific receptor (ETc receptor) from Xenopus laevis dermal melanophores. J Biol Chem 1993;268:19126-33.
25. Simonson MS, Dunn MJ. Cellular signaling by peptides of the endothelin gene family. FASEB J 1990;4:2989-3000.
26. Jouneaux C, Mallat A, Serradeil-Le Gal C, Goldsmith P, Hanoune J, Lotersztajn S. Coupling of endothelin B receptors to the calcium pump and phospolipase C via Gs and Gq in rat liver. J Biol Chem 1994;269:1845-51.
27. Cooper GM, Hausman RE. ŠStanica -molekularni pristupĆ. Medicinska naklada; 2004. (in Croatian)
28. Lotersztajn S. Les endothelines. Med Sci 1993;9:1084-93.
29. Levin ER. Endothelins. N Engl J Med 1995;333:356-63.
30. Housset C, Rockey DC, Bissell DM. Endothelin receptors in rat liver: lipocytes as a contractile target for endothelin-1. Proc Natl Acad Sci USA 1993;90:9266-70.
31. Mallat A, Fouassier L, Préaux AM, Gal CS, Raufaste D, Rosenbaum J, et al. Growth inhibitory properties of endothelin-1 in human hepatic myofibroblastic Ito cells: an endothelin B receptor-mediated pathway. J Clin Invest 1995;96:42-9.
32. Pinzani M, Milani S, De Franco R, Grappone C, Caligiuri A, Gentilini A, et al. Endothelin-1 is overexpressed in cirrhotic liver and exerts multiple effects on activated human hepatic stellate cells. Gastroenterology 1996;110:534-48.
33. Housset C, Carayon A, Housset B, Legendre C, Hannoun L, Poupon R. Endothelin-1 secretion by human gallbladder epithelial cells in primary culture. Lab Invest 1993;69:750-5.
34. Kojima H, Sakurai S, Kuriyama S, Yoshiji H, Imazu H, Uemura M, et al. Endothelin-1 plays a major role in portal hypertension of biliary cirrhotic rats through endothelin receptor subtype B together with subtype A in vivo. J Hepatol 2001;34:805-11.
35. Oshita M, Takei Y, Kawano S, Yoshihara H, Hijioka T, Fukui H, et al. Roles of endothelin-1 and nitric oxide in the mechanism for ethanol-induced vasoconstriction in rat liver. J Clin Invest 1993;91:1337-42.
36. Serradeil-Le Gal C, Jouneaux C, Sanchez-Bueno A, Raufaste D, Roche B, Préaux AM, et al. Endothelin action in rat liver. Receptors, free Ca2+ oscillations, and activation of glycogenolysis. J Clin Invest 1991;87:133-8.
37. Rockey DC, Chung JJ. Inducible nitric oxide synthase in rat hepatic lipocytes and the effect of nitric oxide on lipocyte contractility. J Clin Invest 1994;95:1199-206.
38. Shah V, Toruner M, Haddad F, Cadelina G, Papapetropoulos A, Choo K, et al. Impaired endothelial nitric oxide synthase activity associated with enhanced caveolin binding in experimental cirrhosis in the rat. Gastroenterology 1999;117:1222-8.
39. Dai DZ, Dai Y. Role of endothelin receptor A and NADPH oxidase in vascular abnormalities. Vasc Health Risk Manag 2010;6:787-94.
40. Erbağcı AB, Deveci R, Akarsu E, Tiryaki Ö, Aksoy SN. Efect of intensive insulin therapy on systemic nitric oxide levels and adenosine deaminase activity in secondary sulfonylurea failure. Biochem Medica 2009;19:166–76.
41. Matsuo J, Oku H, Kanbara Y, Kobayashi T, Sugiyama T, Ikeda T. Involvement of NADPH oxidase and protein kinase C in endothelin-1-induced superoxide production in retinal microvessels. Exp Eye Res 2009;89:693-9.
42. Vatter H, Konczalla J, Seifert V. Endothelin related pathophysiology in cerebral vasospasm: what happens to the cerebral vessels? Acta Neurochir Suppl 2011;110:177-80.
43. Lipa JE, Neligan PC, Perreault TM, Baribeau J, Levine RH, Knowlton RJ, et al. Vasoconstrictor effect of endothelin-1 in human skin: Role of ETA and ETB receptors. Am J Physiol 1999;276:H359–67.
44. Freixa X, Heras M, Ortiz JT, Argiró S, Guasch E, Doltra A, et al. Usefulness of endothelin-1 assessment in acute myocardial infarction. Rev Esp Cardiol. 2011;64:105-10.
45. Ikeda T, Ohta H, Okada M, Kawai N, Nakao R, Siegl PK, et al. Antihypertensive effects of a mix edendothelin-A-and-B-receptor antagonist, J-104132, were augmented in the presence of an AT1-receptor antagonist, MK-954. J Cardiovasc Pharmacol 2000;36:S337–41.
46. Schiffrin EL. Role of endothelin-1 in hypertension and vascular disease. Am J Hypertens 2001;14:83S–9S.
47. Jasmin JF, Lucas M, Cernacek P, Dupuis J. Effectiveness of a nonselective ETA/B and a selective ETA antagonist in rats with monocrotaline induced pulmonary hypertension. Circulation 2001;103:314–8.
48. Rubin LJ, Badesch DB, Barst RJ, Galie N, Black CM, Keogh A, et al. Bosentan therapy for pulmonary arterial hypertension. N Engl J Med 2002;346:896-903.
49. Woo SH, Lee CO. Effects of endothelin-1 on Ca2+ signaling in guinea-pig ventricular myocytes: role of proteinkinase C. J Mol Cell Cardiol 1999;31:631–43.
50. Komati H, Maharsy W, Beauregard J, Hayek S, Nemer M. ZFP260 an inducer of cardiac hypertrophy and a nuclear mediator of endothelin-1signaling. J Biol Chem 2011;286:1508–16.
51. Abraham DJ, Krieg T, Distler J, Distler O. Overview of pathogenesis of systemic sclerosis. Rheumatology (Oxford) 2009;48(l3):iii3–7.
52. Wynn TA. Common and unique mechanisms regulate fibrosis in various fibroproliferative diseases. J Clin Invest 2007;117:524–9.
53. Shi-Wen X, Rodríguez-Pascual F, Lamas S, Holmes A, Howat S, Pearson JD, et al. Constitutive ALK5-independent c-Jun N-terminal kinase activation contributes to endothelin-1 overexpression in pulmonary fibrosis: evidence of an autocrine endothelin loop operating through the endothelin A and B receptors. Mol Cell Biol 2006;26:5518–27.
54. Korn JH, Mayes M, Matucci Cerinic M, Rainisio M, Pope J, Hachulla E, et al. Digital ulcers in systemic sclerosis: prevention by treatment with bosentan, an oral endothelin receptor antagonist. Arthritis Rheum 2004;50:3985–93.
55. Sulli A, Soldano S, Pizzorni C, Montagna P, Secchi ME, Villaggio B, et al. Raynaud’s phenomenon and plasma endothelin: correlations with capillaroscopic patterns in systemic sclerosis. J Rheumatol 2009;36:1235–9.
56. Widyantoro B, Emoto N, Nakayama K, Anggrahini DW, Adiarto S, Iwasa N, et al. Endothelial cell-derived endothelin-1 promotes cardiac fibrosis in diabetic hearts through stimulation of endothelial-to-mesenchymal transition. Circulation 2010;121:2407–18.
57. Edwin Wu, Maria MIG. Cardiac magnetic resonance imaging and endothelin-1: A step forward into the detection of microvascular obstruction. Rev Esp Cardiol 2011;64:89-91.
58. Eriksson C, Gustavsson A, Kronvall T, Tysk C. Hepatotoxicity by Bosentan in a Patient with Portopulmonary Hypertension: a Case-Report and Review of the Literature J Gastrointestin Liver Dis 2011;20:77-80.
59. Kosanovic D, Kojonazarov B, Luitel H, Dahal BK, Sydykov A, Cornitescu T, et al. Therapeutic efficacy of TBC3711 in monocrotaline-induced pulmonary hypertension. Respir Res 2011;12:87.
60. Frampton JE. Ambrisentan. Am J Cardiovasc Drugs 2011.
61. Macdonald RL, Higashida RT, Keller E, Mayer SA, Molyneux A, Raabe A, et al. Clazosentan, an endothelin receptor antagonist, in patients with aneurysmal subarachnoid haemorrhage undergoing surgical clipping: a randomized, double-blind, placebo-controlled phase 3 trial (CONSCIOUS-2). ) Lancet Neurol 2011;10:618-25.
62. Clarkson-Jones JA, Kenyon AS, Tomkinson HK. The disposition and metabolism of zibotentan (ZD4054): an oral-specific endothelin A receptor antagonist in mice, rats and dogs. Xenobiotica 2011;41:784-96.
63. O’Callaghan DS, Savale L, Yaïci A, Natali D, Jaïs X, Parent F, et al. Endothelin receptor antagonists for the treatment of pulmonary arterial hypertension. Expert Opin Pharmacother. 2011;12:1585-96.
64. Dwyer N, Kilpatrick D. Bosentan for the treatment of adult pulmonary hypertension. Future Cardiol 2011;7:19-37.
65. Jeremy JY, Shukla N, Angelini GD, Wan S. Endothelin-1 (ET-1) and vein graft failure and the therapeutic potential of ET-1 receptor antagonists. Pharmacol Res 2011;63:483-9.
66. Dhaun N, Goddard J, Kohan DE, Pollock DM, Schiffrin EL, Webb DJ. Role of Endothelin-1 in Clinical Hypertension - 20 Years On. Hypertension 2008;52:452-9.
67. Weber MA, Black H, Bakris G, Krum H, Linas S, Weiss R, et al. A selective endothelin-receptor antagonist to reduce blood pressure in patients with treatment-resistant hypertension: a randomised, double-blind, placebo-controlled trial. Lancet 2009;374:1423–31.
68. Epstein BJ. Efficacy and Safety of Darusentan: A Novel Endothelin Receptor Antagonist. Ann Pharmacother 2008;42:1060-9.
69. Rossi GP, Colonna S, Pavan E, Albertin G, Della Rocca F, Gerosa G, et al. Endothelin-1 and its mRNA in the wall layers of human arteries ex vivo. Circulation 1999;99:1147–55.
70. Roudrigez-Pascal F, Busnadiego O, Lagares D, Lamas S. Role of endothelin in the cardiovascular system. Pharmacol Res 2011; 63:463-72.
71. Simeone SM, Li MW, Paradis P, Schiffrin EL. Vascular gene expression in mice overexpressing human endothelin-1 targeted to the endothelium. Physiol Genomics 2011;43:148–60.
72. Barton M, Haudenschild CC, d’Uscio LV, Shaw S, Munter K, Luscher TF. Endothelin ETA receptor blockade restores NO-mediated endothelial function and inhibits atherosclerosis in apolipoprotein-E-deficient mice. Proc Natl Acad Sci USA 1998;95:14367-72.
73. Suhail M, Faizul-Suhail M. Maternal and cord blood malondialdehyde and antioxidant vitamin levels in normal and preeclamptic women. Biochem Medica 2009;19:182–91.
74. Dechanet C, Fort A, Barbero-Camps E, Dechaud H, Richard S, Virsolvy A. Endothelin-dependent vasoconstriction in human uterine artery: application to preeclampsia. PLoS One 2011;6:e16540.
75. Said N, Smith S, Sanchez-Carbayo M, Theodorescu D. Tumor endothelin-1 enhances metastatic colonization of the lung in mouse xenograft models of bladder cancer. J Clin Invest 2011;121:132-47.
76. Nabhan C, Parsons B, Touloukian EZ, Stadler WM. Novel approaches and future directions in castration-resistant prostate cancer. Ann Oncol 2011. ŠEpub ahead of printĆ
77. Bagnato A, Loizidou M, Pflug BR, Curwen J, Growcott J. Role of the endothelin axis and its antagonists in the treatment of cancer. Br J Clin Pharmacol 2011;163:220-33.
78. Stern PH, Tatrai A, Semler DE, Lee SK, Lakatos P, Strieleman PJ, et al. Endothelin receptors, second messengers and actions in bone. J Nutr 1995;125:2028S-32S.
79. Geneau G, Lamiche C, Niger C, Strale PO, Clarhaut J, Defamie N, et al. Effect of endothelin-1 on osteoblastic differentiation is modified by the level of connexin43: comparative study on calvarial osteoblastic cells isolated from Cx43+/- and Cx43+/+ mice. Cell Tissue Res 2010;340:103-15.
80. Ruest LB, Clouthier DE. Elucidating timing and function of endothelin-A receptor signaling during craniofacial development using neural crest cell-specific gene deletion and receptor antagonism. Dev Biol 2009;328:94-108.
81. Sato T, Kurihara Y, Asai R, Kawamura Y, Tonami K, Uchijima Y, et al. An endothelin-1 switch specifies maxillomandibular identity. Proc Natl Acad Sci U S A 2008;105:18806-11.
82. Gertz ER, Silverman NE, Wise KS, Hanson KB, Alekel DL, Stewart JW, et al. Contribution of Serum Inflammatory Markers to Changes in Bone Mineral Content and Density in Postmenopausal Women:A 1-Year Investigation. J Clin Densitom 2010;13:277–82.
83. Imhof A-K, Glück L, Gajda M, Bräuer R, Schaible HG, Schulz S. Potent antiinflammatory and antinociceptive activity of the endothelin receptor antagonist bosentan in monoarthritic mice. Arthritis Res Ther 2011;13:R97.
84. Gossl M, Lerman A. Endothelin beyond a vasoconstrictor. Circulation 2006;113:1156-8.