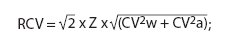Introduction
Cardiac troponin concentration is the most useful biomarker for suspected acute coronary syndrome (ACS) management in (ED). The troponin assay configuration, featuring a combination of antibodies against the heart-specific and stable region of the molecule, determines their clinical performance. However, the interpretation of test results requires not only a comparison with a reference interval or threshold value, but also a comparison between consecutive measurements (1). International recommendations of Cardiology Society and Laboratory Medicine established that the Tn-I decision limit should be at the 99thpercentile value of a reference group with a total analytical imprecision (as coefficient of variation) ≤ 10 % (2). Actually, few studies have considered a longitudinal comparison, as estimated by the change between two consecutive troponin tests. We investigated this specific point by analysing the variations of troponin-I in two consecutive tests from the same patient, as obtained in the laboratory routine practice.
This work aimed at: a) estimating the biological and analytical variation of plasma cardiac troponin I (cTn-I) measurement for a specific analytical system, to be used for the reference change value (RCV) calculation; b) assessing the diagnostic accuracy of the RCV of cTn-I for diagnosing the ACS in patients with second elevation of cTn-I after 4-6 hours.
Materials and methods
Patients and analytical methods
From the laboratory database we collected data from 103 patients (age between 31 and 93 years, median age 72) admitted to the cardiological ED in a period of 9 months. Patients inclusion criteria were based on the cTn-I increases in the ED with the first value of the cTn-I < 0.1 µg/L, referred to consecutive patients admitted for chest pain. Final diagnosis was made at the end of clinical evaluation, considering medical, biochemical and electrocardiogram/angiographic analysis by an expert medical staff. From the emergency room patients without acute coronary syndrome and/or other acute pathologies were discharged. Patients with ACS or other clinical situations justifying troponin increases (i.e. cardiac heart failure or pulmonary edema) were hospitalised. The patients who underwent coronary examination and/or final medical diagnosis were divided into four groups: 1) patients with severe coronaropathy, ST-segment elevation myocardial infarction (STEMI), 2) patients with frequently mono-bivascular coronaropathy, non-ST-segment elevation myocardial infarction (NSTEMI), 3) patients with cardiac heart failure or pulmonary edema, 4) patients with atrial fibrillation or tachycardia. The diagnostic criteria for ACS and subgroups were defined according to ACC/AHA 2007 guidelines and to good clinical practice of expert cardiologist team. All the patients included in the study were admitted to ED within 6 hours from the onset to the first blood sample, approximately. According to the procedures of our ED, blood samples for testing were collected at admission and after 4-6, 12, 18, 24 hours. In our study the two first consecutive cTn-I values were evaluated in order to interpret the early signs of biochemical events. The second result was recorded as significant when the variation (increase) of cTn-I concentration exceeded the critical value, calculated as shown below. The 103 clinical cases were divided into 4 categories according to the difference between two consecutive cTn-I measurements (significant or not) and to the presence of confirmed ACS (clinically confirmed or not). A 2 way contingency table was constructed in order to assess the association between the increased cTn-I values and the ACS. All cTn-I measurements were performed with the Access system (DXI Unicel 800 and Access I, Beckman Coulter), using the Accu troponin-I (cTn-I) assay (Beckman Coulter) reagents and calibrators kit, following the manufacturer’s instructions.
Biological variation
70 healthy individuals (41 women and 29 men; healthy state assessed by medical evaluation and laboratory tests) were enrolled to evaluate within-subject (CVw) and between-subject (CVbt) biological variation of cardiac troponin-I. Ten subjects out of 70 were retested during the period of study, under routine analytical conditions and according to suggested protocol, to estimate the CVw (3,4). During a period of six months, three blood samples, randomly in different working day, were drawn and each subject assayed. According to preliminary data, we reputed sufficiently accurate the protocol for CVw evaluation with 10 subjects tested (5).
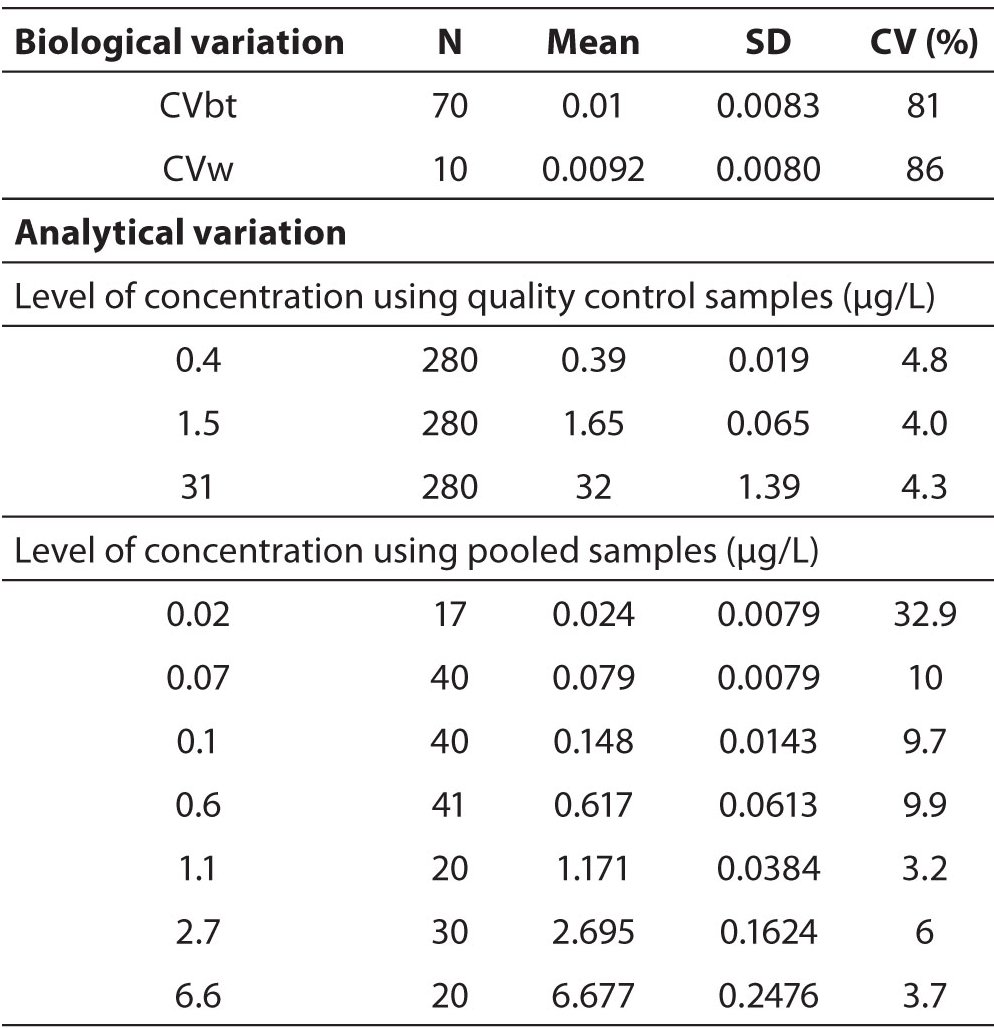
The cTn-I CVw, as well as the cTn-I CVbt of the population, was calculated as coefficient of variation (CV) obtained by the cTn-I measurements of the healthy subjects (Table 1). The CV was calculated as the ratio of the standard deviation to the mean of all the cTn-I measurements.
Table 1. Sources of variability of the cTn-I measurements for cTn-I RCV calculation. It’s very high variation close to the detection limit of 0.01 µg/L.
Analytical variation
The analytical variation was calculated as the coefficient of variation observed during 9 months of internal quality control of the analytical system, at three concentration levels, and including control charts monitoring and application of control rules (Table 1). In order to further evaluate the analytical imprecision of the analyser, we have performed an imprecision profile study (Figure 1). A pool of 32 plasma samples were used to prepare 7 pools with cTn-I concentrations ranging from 0.02 to 6 µg/L. They were stored at -30 °C until measurement, when they were thawed, equilibrated to room temperature, and centrifuged before measurement. We then performed repeated measurements of each aliquot per day in a period of about 3 months. Finally, the total coefficient of variation was calculated for each cTn-I concentration (Table 1).
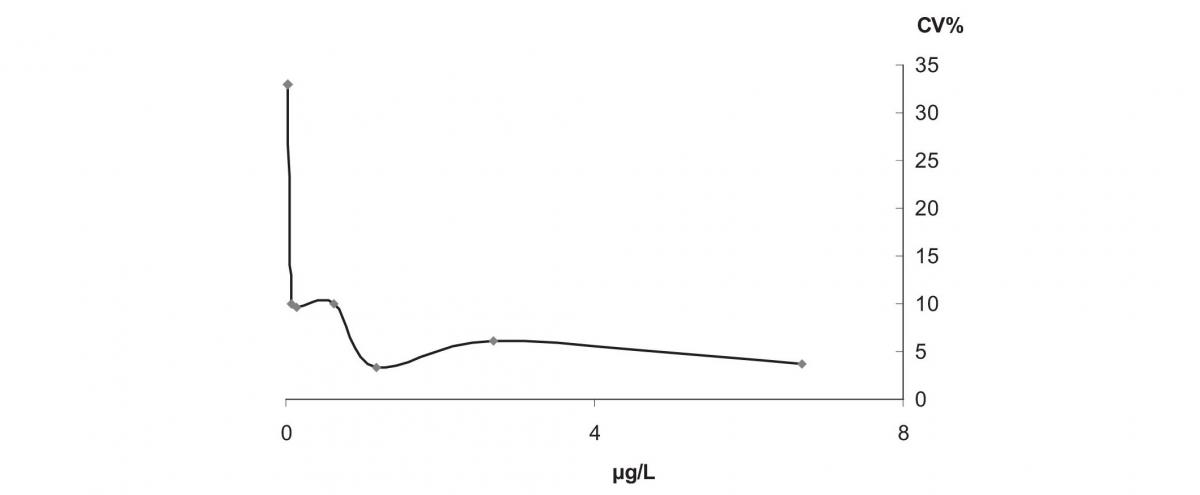
Figure 1. Imprecision profile of cTn-I measurement obtained by our analytical system. For concentrations close to the analytical sensitivity limit (0.01 µg/L) the total CV becomes very high.
Preanalytical conditions
Blood samples with heparin as anticoagulant were collected, centrifuged at 2500 x g for 10 minutes, and analysed freshly. Adequate filling of the collection tube and immediate mixing after collection were assured. Haemolysed samples with free haemoglobin > 500 mg/dL were excluded.
Statistical evaluation
Elaboration of laboratory and clinical data was made using Microsoft Office Excel 2003, considering clinical cases with the first measurement of cTn-I < 0.1 µg/L. Biochemical measurements of cTn-I increases were presented as median value and interquartile range (IQR). Data referred to the analytical and biological variation estimation were illustrated as mean and standard deviation. The difference between cTn-I RCV values for patients hospitalised and for patients discharged was performed using Chi square test through SPSS statistics 17.0 Version. As derived quantities of Chi square test we calculated the NPV and PPV of the RCV parameter. The level of statistical significance (P) accepted was < 0.05.
RCV and the cTn-I critical value RCV
The RCV for two consecutive values of cTn-I was calculated according to Fraser and Harris’ formula (6,7):
where Z is the z-statistic equal to 1.96 at 95% statistical significance, CVw is the within-subject biological variation and CVa is the analytical variation.
In order to assess the significance of possible differences between two consecutive results from the same patient, we have introduced “critical value of troponin RCV” (CrRCV) calculated as follows:
CrRCV = [first cTn-I result] + [(first cTn-I result) x RCV];
where CV and CrRCV are expressed in unit of concentration (µg/L). This approach allows an immediate evaluation of the significance of any difference between two measured consecutive values of cTn-I: when the second concentration value is higher than the CrRCV, any observed increase is statistically significant.
Results
We found a within-subject biological variation CVw equal to 86% as coefficient of variation of the 30 cTn-I measurements obtained from 10 healthy subjects; the analytical variation was 4.4%, at the concentration level of 0.4 µg/L and for higher levels (see Table 1); from these we estimated a RCV equal to 240%. This result was due to the high analytical variation of cTn-I measurement at the low concentrations, close to the analytical detection limit 0.01 µg/L, of the majority of healthy individuals. Consequently, we justified an overestimation of the cTn-I biological variation measure. The cTn-I imprecision profile (Figure 1)showed that at 0.02 µg/L the analytical variation as total CV was 32%, while, the 10% of CV was reached at 0.07 µg/L, close to the decision limit for myocardial injury (equal to 0.05 µg/L, corresponding to the 99th percentile of a reference population).
Theincreased cTn-I concentrations for patients admitted to ED for the suspect of ACS and confirmed through clinical and angiographic data were clinically significant.
The initial concentration value at admission to ED, for patients discharged after clinical evaluation, was 0.015 µg/L (median value) with a maximum of 0.08 µg/L. This means that some cTn-I values clinically negative can fall close to the decision limit of 0.05 µg/L in the suspect of ACS.
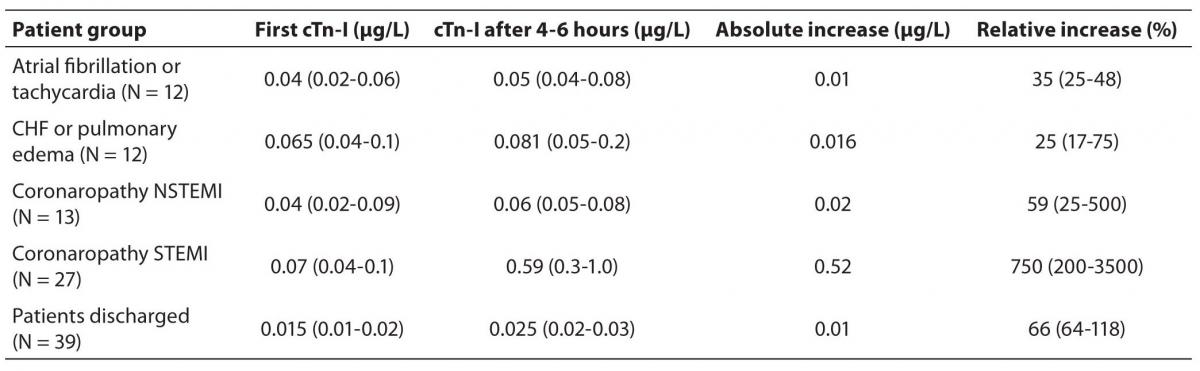
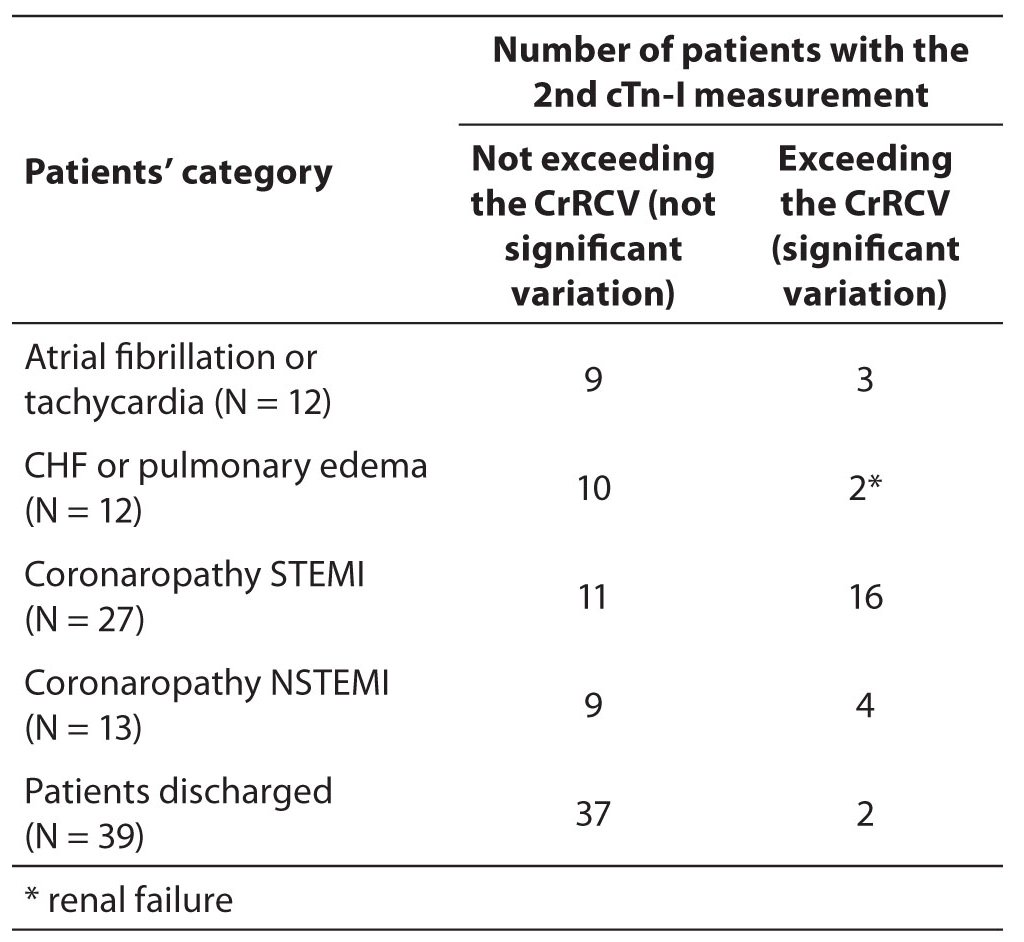
From the laboratory database we have selected data with the first cTn-I result ≤ 0.1 µg/L, then a comparison of the second result with the CrRCV was assessed. As derived quantities of Chi square analysis, we have estimated the cTn-I RCV negative predictive value (NPV) equal to 88% with confidence interval at 95% (CI = 82-92%). The positive predictive value (PPV) for cTn-I RCV was 52% with (CI = 37-64%). The sensitivity was 62%, (CI = 44-77%) and the specificity 83%, (CI = 77-87%). The application of cTn-I RCV critical value to the two consecutive results from 103 patients admitted to ED is shown in Table 3. We found that the cTn-I RCV was effectiveness for the detection of some ACS cases; conversely, it can exclude, with high probability, the not significant cTn-I increases. We found that patients suffering from severe coronaropathies exhibited a high percentage of significant increase of cTn-I between two consecutive results (Table 2).
Table 2. Amount of plasma cTn-I increases in pair of consecutive measurements. Figures are the concentrations and cTn-I increases expressed as median value and IQR. Plasma cTn-I increase, 2nd over 1st sample.
Table 3. Cases with increased plasma cTn-I concentration in the 2nd measurement, exceeding or not exceeding the CrRCV (P < 0.001). Categories reflect the final diagnosis in patients admitted for chest pain in cardiological ED.
The most consistent result concerns the cTn-I increase of the group of patients discharged. We have shown that many patients with low cTn-I increases and not exceeding the cTn-I RCV were discharged after clinical evaluation (Table 3). The difference between cTn-I RCV values for patients admitted to hospital with a final diagnosis and values for patients discharged from ED was statistically significant (Chi square P < 0.001).
Discussion
Cardiac troponins I or T are commonly used in clinical laboratory as biomarkers of myocardial necrosis. In an experimental comparison study, the clinical performance of Accu troponin-I (cTn-I) assay (Beckman Coulter) has been assessed (8). The AccuTn-I assay, based on a pair of monoclonal antibodies against epitopes close to the NH2 terminus (epitopes 24-40 and 41-49), actually shows a good sensitivity and specificity for acute coronary syndrome. Commercial assays and analytical systems are available from the diagnostic manufacturers with improved test sensitivity and analytical reliability; however, it is well understood that the future generations of assays for cardiac troponin should improve the sensitivity of biomarkers detection (9,10). In the last period, several studies in progress have the aim to assess diagnostic assays with ultrasensitive cTnI, controlling the analytical specificity. The technology-dependent sensitivity limits or unrecognizable interferences might cause misleading laboratory report (11). The interpretation of laboratory tests remains important to evaluate and quantify the myocardial injury in order to improve the prognosis of the patients (12). In particular, the longitudinal comparison of biomarker results in serial determinations is not presently given enough attention in clinical laboratory routine as well as the reliable statistical measure of reference population (13,14). In the present observational retrospective study, we have applied the CrRCV, a new RCV derived parameter, to interpret the second of two consecutive cTn-I measurement results in the evaluation of clinical cases in ED. To investigate the clinical effectiveness of cTn-I RCV, calculated in our laboratory, we have analysed the retrospective cTn-I increases in different cardiological pathologies. We have considered low elevations of cTn-I results even with concentrations below the 99th percentile of the reference population. In fact, recent studies have demonstrated that minor elevations of cardiac troponins are clinically significant for cardiovascular events as well as elevated troponin levels (15,16).
Patients admitted for chest pain to ED revealed that many cTn-I increases can have a casual fluctuation that could determine a misclassification of patients. In fact, the cTn-I RCV showed a high negative predictive value with a statistically significant association with the exclusion of ACS in patients in ED (see Table 3). However, in AMI or severe coronaropathy cases, the short-term increased cTn-I concentration, greater than cTn-I RCV, could better evaluate the biochemical event of the myocardial necrosis. On the other hand, the calculation methodology of cTn-I RCV introduces a tool to quantify the imprecision and analytical noise of immunoassay technique for low troponin concentrations. According to our finding, a multicenter study conducted on the commercially available assays has demonstrated, with the same experimental protocol, a high cTn-I imprecision at low concentration ranges (17). These investigations suggest that the analytical and biological variability of cardiac troponin-I should be quantified, and the reference change value should be applied as indicator of the significance of the change between two consecutive values (18). Nevertheless, our laboratory results suggest that they may not be applied extensively (without extrapolation to the general population), but they are valuable and deserve further investigation. The cTn-I RCV calculated, could be applied in medical routine only for low cTn-I levels (< 0.1 µg/L), and for a specific immunoassay technique, because of its intrinsic imprecision (17). In the evaluation of cTn-I levels around the decision limit of 0.05 µg/L, non significant plasma variations could be detected and interpreted by comparison with the cTn-I RCV. In fact, the high NPV of cTn-I RCV suggests its possible use to aid the rule out of patients in ED with minor not specific cTn-I elevations. We focused on the features of the immunochemical method that determine a high analytical imprecision of biomarker, close to the detection limit of 0.01 µg/L, with a contribution to high values of biological variability and RCV. We recognised the limitations of our study, also, in regard of the RCV calculation and sensitivity of the analytical method routinely used. An additional adverse effect of low sensitivity of method makes the RCV estimates method-dependent. In practice, a lower detection limit of cTn-I assay could improve the within-subject biological variation estimation, which the RCV calculation is based upon. In conclusion, we admitted limitations both in regard of the homogeneous population of the clinical cases due to a lack of baseline comparability, and to the poor evidences to distinguish among the possible forms of association. However, the diagnostic accuracy was done retrospectively and surely might have introduced a certain amount of overestimation. In any case, a reference change applied to a longitudinal analysis of cardiac biomarkers remains a challenge that requires further clinical and prospective studies.
Acknowledgements
We would like to thank the technical staff for his collaborative work. Professor C. Franzini and V. Biagioli assisted in the preparation and revision of the manuscript.
Notes
Potential conflict of interest
None declared.
References
1. Ricos C, Domenech MV, Perich C. Analytical quality specifications for common reference intervals. Clin Chem Lab Med 2004;42:856-62.
2. The joint European society of cardiology/American College of cardiology committee. Myocardial infarction redefined-A consensus document of the joint European society of cardiology/American College of cardiology committee for the redefinition of myocardial infarction. JACC 2000;36:959-69.
3. Harris EK. Effect of intra-and inter-individual variation on the appropriate use of normal ranges. Clin Chem 1974;20:1535-42.
4. Petersen PH, Sandberg S, Fraser CG, Goldschmidt H. Influence of index of individuality on false positives in repeated sampling from healthy individuals. Clin Chem Lab Med 2001;39:160-5.
5. Ricos C, Perich C, Minchinela J, Alvarez V, Simon M, Biosca C, et al. Application of biological variation - a review. Biochem Med 2009;19:250-9.
6. Fraser CG. Biological Variation from Principles to Practice, AACC press, Washington 2001.
7. Harris EK, Yasaka T. On the calculation of ‘’reference change’’ for comparing two consecutive measurements. Clin Chem 1983;29:25-30.
8. James S, Flodin M, Johnston N, Lindahl B, Venge P. The antibody configuration of cardiac troponin I assay may determine their clinical performance. Clin Chem 2006;52:832-7.
9. NACB writing group members, Morrow DA, et al. NACB committee members Christenson RH, et al. National academy of clinical biochemistry, laboratory medicine practice guidelines: clinical characteristics and utilization of biochemical markers in acute coronary syndromes. Clin Chem 2007;53:552-74.
10. Casals G, Filella X, Bedini JL. Evaluation of a new ultrasensitive assay for cardiac troponin I. Clin Biochem 2007; 40:1406-13.
11. Dodig S. Interferences in quantitative immunochemical methods. Biochem Med 2009;19:50-62.
12. Parenti N, Bartolacci S, Carle F, Angelo F. Cardiac troponin as prognostic marker in heart failure patients discharged from emergency department. Intern Emerg Med 2008;3:43-7.
13. Ricos C, Cava F, Garcia-Lario JV, Hernandez A, Iglesias N, Jimenez CV, et al. The reference change value: a proposal to interpret laboratory reports in serial testing based on biological variation. Scand J Clin Lab Invest 2004;64:175-84.
14. Simundic AM. Confidence interval. Biochem Med 2008;18:154-61.
15. Thielmann M, Massoudy P, Neuhauser M, Tsagakis K, Margggraf G, Kamler M, et al. Prognostic value of preoperative cardiac troponin I in patients undergoing emergency coronary artery bypass surgery with non-elevation or ST-elevation acute coronary syndromes. Circulation 2006;114:I448-53.
16. Kavsak PA, Newman AM, Lustig V, MacRae AR, Palomaki GE, Ko DT, et al. Long-term health outcomes associated with detectable troponin I concentrations. Clin Chem 2007;53:220-7.
17. Panteghini M, Pagani F, Yeo K-Teck J, Apple FS, Chrstenson RH, Dati F, et al. Evaluation of Imprecision for cardiac troponin assays at low-range concentrations. Clin Chem 2004;50:327-32.
18. Smellie WSA. What is a significant difference between sequential laboratory results? J Clin Pathol 2008;61:419–25.